OUR RAPID RESPONSE TO COVID-19
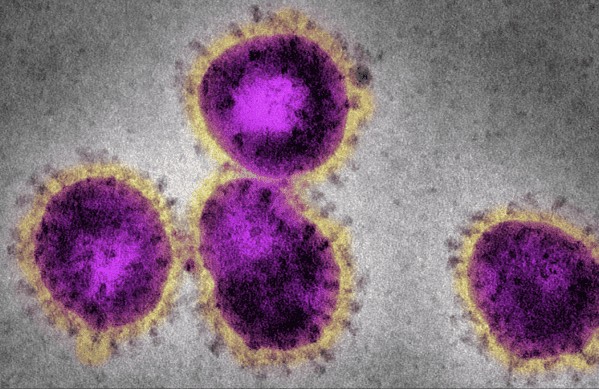
During the COVID-19 pandemic, Regeneron applied our decades of scientific and technology expertise to combat a novel and quickly mutating virus. We were uniquely positioned to face this public health threat given our proprietary VelociSuite® technologies and our track record against infectious diseases such as Ebola.
Our COVID-19-related discovery efforts started in early 2020, when we utilized VelociSuite to produce and evaluate hundreds of virus-neutralizing antibodies in our genetically engineered mice. We also identified similarly performing antibodies from human COVID-19 survivors in order to maximize the pool of potential candidates. We quickly evaluated and selected two potent, complementary antibodies that bound to different, non-overlapping parts of the spike protein of the SARS-CoV-2 virus, thereby blocking the virus’s ability to infect healthy cells.
Improving on our prior infectious disease timeline, this combination was ready to enter human clinical trials just five months after the world first learned the SARS-CoV-2 viral sequence. As part of our rapid response approach and in recognition of the tremendous global need, we simultaneously initiated clinical trials and our large-scale manufacturing process. Manufacturing biologic medicines like monoclonal antibody therapies is a time-consuming and precise process, and unfortunately biology can’t be hurried along. Despite the risks and uncertainties, we purposefully began this effort as early as possible so that we would have as many doses as possible immediately available for people around the world.
Helping patients in a time of need
Our investigational antibody medicine combination received emergency use authorization (EUA) from the U.S. Food and Drug Administration in November of 2020, the first combination therapy to receive an EUA and in record time – only 10 months after the program's inception.
The U.S. government signed multiple agreements with Regeneron for the purchase of nearly three million doses of the investigational antibody therapy and made it available for free to patients who qualified under the EUA criteria. We also collaborated with Roche to increase global supply of our medicine, with Regeneron responsible for development of the therapy in the U.S., and Roche primarily responsible for development and distribution outside the U.S. Our companies made a shared commitment to making the antibody combination available to COVID-19 patients around the globe and to supporting access in low- and lower-middle-income countries through drug donations to be made in partnership with public health organizations.
Voluntary Revocation of Emergency Use Authorization in the U.S.
Viruses, by their nature, mutate over time leading to variant forms. Indeed, our antibody cocktail remained active against numerous strains of the SARS-CoV-2 virus, including Delta; however, it lost potency against the Omicron variant. Today, the antibody cocktail is not currently authorized for use in any U.S. states, territories or jurisdictions.
In January 2022, the FDA amended the EUA to exclude its use in geographic regions where infection or exposure is likely due to a variant that is not susceptible to the treatment. In November 2024, we voluntarily submitted a request to the FDA for the revocation of the EUA. Please refer to this statement on our voluntary revocation of the EUA.
Ongoing research in infectious diseases
Regeneron’s infectious disease programs have led to an approved medicine for Ebola, an emergency use authorized medicine for COVID-19, and an investigational medicine for Middle East Respiratory Syndrome (MERS). We continue to explore new potential medicines for emerging infectious diseases and established viruses like influenza, in part through long-standing research collaborations with BARDA.
Learn how we applied our homegrown technologies and novel scientific approach to 2014's West African Ebola outbreak.
From discovery to large-scale manufacturing, our VelociSuite® technologies uniquely enable our discovery and development efforts.